UCL-3500紫外交聯儀在紫外線誘導突變中的應用
摘要:
2025 年,北京化工大學化學工程學院聯合生命科學與技術學院綠色生物制造國家重點實驗室在《Biochemical Engineering Journal》期刊發表文獻《Screening of high-yielding β-farnesene Saccharomyces cerevisiae strains and quantification of fermentation processes: A new fluorescence-based high-throughput screening and quantification platform》,該文獻明確指出利用紫外交聯儀 UCL-3500 可高效開展紫外線誘導突變實驗,為高產 β- 法尼烯釀酒酵母菌株的篩選及發酵過程量化提供了關鍵技術支撐,顯著提升了紫外線誘導突變篩選的效率與準確性。
紫外交聯儀 UCL-3500 助力紫外線誘導突變篩選高產酵母菌株
2025 年,北京化工大學主導的研究團隊在《Biochemical Engineering Journal》發表重要成果,成功建立了基于熒光探針的高通量篩選與定量平臺,實現了從紫外線誘導突變菌株篩選到規模化發酵過程監測的全流程優化。在該研究中,紫外交聯儀 UCL-3500 作為核心實驗設備,承擔了釀酒酵母的紫外線誘導突變任務,其穩定的紫外線輸出、精準的照射時間控制能力,為構建高質量突變體庫、高效篩選高產 β- 法尼烯菌株奠定了堅實基礎。
北京化工大學化學工程學院與綠色生物制造國家重點實驗室是我國生物化工領域的重要研究陣地,圍繞綠色可持續生產的重大戰略需求,在微生物代謝工程、生物催化與轉化、發酵過程優化等前沿領域開展系統研究。重點方向包括萜類化合物生物合成、微生物菌株改良、高效檢測技術開發等,曾在多個國際優秀期刊發表多項突破性成果,為生物制造產業的升級發展提供了重要理論支撐和技術儲備。
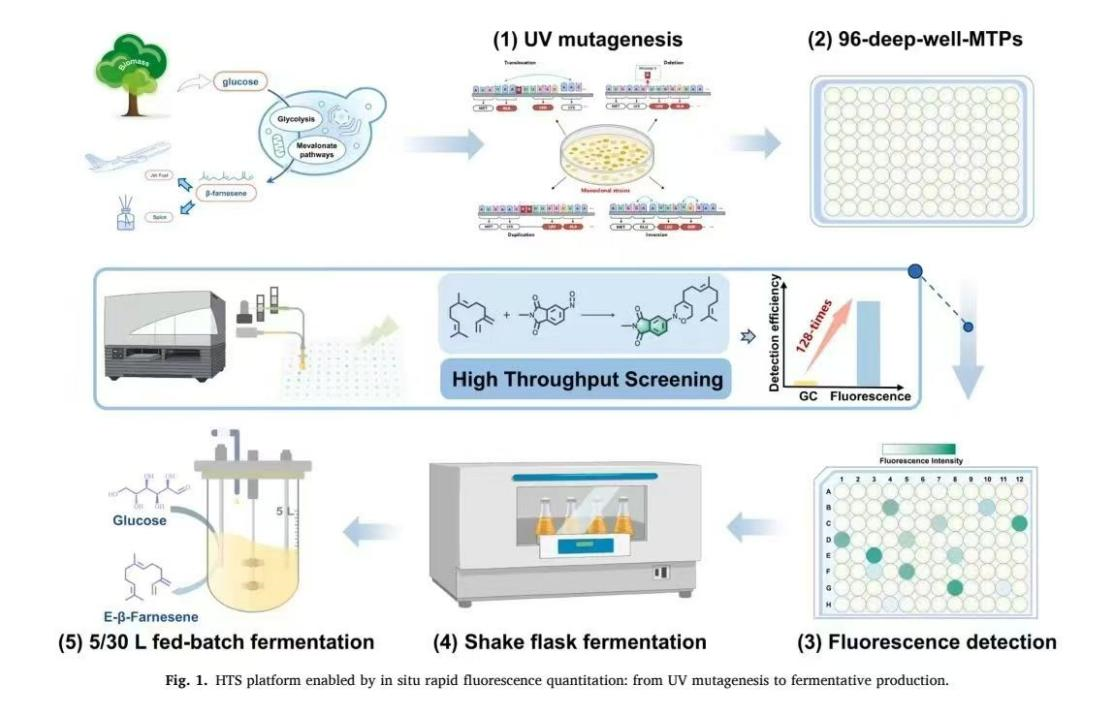
在該實驗中,研究團隊以產 β- 法尼烯工程釀酒酵母菌株 BUHY0003 為研究對象,采用紫外交聯儀 UCL-3500 開展紫外線誘導突變實驗。研究人員將 5mL 菌株懸液置于無菌培養皿中,加入無菌磁力攪拌子使菌液形成均勻薄層,隨后將培養皿置于紫外交聯儀 UCL-3500 紫外線燈下方約 20cm 處,設置 0、20、25、30 等多個照射時間梯度進行紫外線誘導突變處理,每個處理組均設置三次重復。紫外線誘導突變處理后,將菌液在黑暗條件下離心收集,重懸于 YPD 培養基中恢復培養 2 小時,再涂布于 YPD 瓊脂培養基上培養,通過計算菌落存活率確定紫外線誘導突變參數。最終發現,當紫外交聯儀 UCL-3500 的照射時間為 50 秒時,菌株致死率達到 94.76%,此條件下既能保證足夠的突變負荷,又能維持突變體庫的實用規模,是開展紫外線誘導突變的設置。
通過紫外交聯儀 UCL-3500 完成紫外線誘導突變后,研究團隊利用構建的熒光高通量篩選平臺對突變菌株進行篩選,成功獲得高產突變株 BUHY0004,其 β- 法尼烯產量較原始菌株提升 37.34%。后續經 5L 和 30L 補料分批發酵驗證,該突變株表現出良好的規模化生產性能,產量分別達到 35.1g/L 和 33.3g/L,充分證明了紫外交聯儀 UCL-3500 在紫外線誘導突變菌株改良中的可靠性與高效性。
紫外交聯儀 UCL-3500 是一款專為微生物紫外線誘導突變設計的專業設備,采用穩定的紫外線光源設計,可提供均勻、高強度的紫外線照射,確保紫外線誘導突變的有效性。設備支持靈活的照射時間調節與能量調控,能滿足不同菌株的紫外線誘導突變需求,可精準控制突變強度,為篩選有益突變體創造有利條件。操作簡便,無需復雜的前期調試,適用于實驗室大規模突變體庫構建。其適配性強,可廣泛應用于釀酒酵母、大腸桿菌等多種微生物的紫外線誘導突變實驗,為代謝工程、菌株改良、發酵工業等領域的研究提供強有力的技術支持。目前,該設備已成為高校、科研院所開展微生物紫外線誘導突變相關研究的重要工具,相關研究成果已在多個國際優秀期刊發表。
原文相關段落摘錄:
Saccharomyces cerevisiae is regarded as one of the most important model strain and industrial workhorses in metabolic engineering and biotechnology, and has been extensively employed for the manufacturing of natural products. Although substantial advances in genome editing and rational design have been achieved, growth retardation in scale-up fermentations is frequently observed in engineered yeasts owing to the complex regulation of central carbon metabolism. In this context, ultraviolet (UV) mutagenesis is recognized as a powerful complementary strategy, enabling the rapid generation of extensive mutant libraries and facilitating the identification of beneficial alleles.
UV mutagenesis and 96-well plate screeningIn this study, the engineered S. cerevisiae strain BUHY0003 used for producing β-farnesene was constructed according to our previous work. A mutagenesis chamber (UCL-3500, Shanghai Luyang Biotechnology Co., Ltd., China) equipped with two 18 W UV lamps was used as the light source and was preheated for 20 min before use. 5 mL of BUHY0003 strain suspension were placed in a sterile 9 cm culture dish. A sterile 1.5 cm magnetic stir bar was added, and the dish was gently agitated on a shaker to form a uniform thin layer. The dish was positioned approximately 20 cm below the UV lamps and was irradiated at normal incidence in triplicate. Irradiation times were set to 0, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, and 80 s. Following irradiation, cells were collected by centrifugation at 4000 × g for 4 min in the dark. The supernatant was discarded, and the cells were resuspended in YPD medium followed by a 2-hour recovery incubation at 30?C with shaking at 200 rpm. 100 μL of the suspension were spread onto YPD agar medium and were incubated inverted at 30?C for 96 h. Under sterile conditions, colonies were counted and lethality was calculated.